Food safety is a major societal and industrial issue. In France, according to government statistics, there are around 3,000 product recalls per year, nearly 75% of which have involved food products on average since 2021(1).
Regulations are evolving rapidly to protect consumers. While these efforts are absolutely commendable, it is not always easy for manufacturers to keep up with these changes. This is particularly true for regulations concerning Bisphenol A. We therefore propose to review this topic by revisiting key definitions, assessing the current situation, and taking a closer look at the available packaging solutions.
1- DEFINITIONS
CMR, endocrine disruptors, bisphenols A and others: numerous opinions and toxicological studies have been conducted, are ongoing, and will continue in order to assess their potential toxicity. But are we always clear about their definitions?
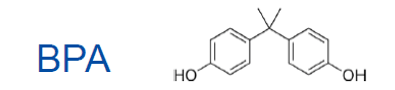
Bisphenol A is “an organic compound from the family of aromatic compounds, mainly used in the manufacture of plastics and resins. Obtained by the reaction between two equivalents of phenol and one equivalent of acetone, its molecule contains two phenol functional groups. It is also referred to as 4,4'-(propan-2-ylidene) diphenol or p,p'-isopropylidenebisphenol.” (2)
More generally, bisphenols constitute a family of chemical substances widely used in the manufacture of everyday plastic products (toys, textiles, coatings of food cans, etc.). While the risks of bisphenol A (BPA) are well established, many other bisphenols used as substitutes for BPA are strongly suspected of also being endocrine disruptors, with serious risks to human health (miscarriages, sexual and reproductive dysfunctions, cognitive delays, etc.).
Endocrine disruptors: the most commonly used definition is that of the WHO in 2002: “An endocrine disruptor is a chemical substance of natural or artificial origin, external to the body, capable of interfering with the functioning of the endocrine system and inducing harmful effects in an intact organism, its offspring, or within a population, including chronic developmental disorders, reproductive issues, hormone-dependent cancers, or metabolic diseases.” They have the particularity of altering dose-response relationships, making them irregular: researchers refer to “non-monotonic dose-response curves,” which makes their evaluation even more difficult.
CMR substances: Carcinogenic – Mutagenic – Reprotoxic
Some chemical agents have, in the medium or long term, carcinogenic, mutagenic, or toxic effects on reproduction. They are referred to as CMR agents.
Carcinogenic agent or process: a chemical product (asbestos, wood dust, benzene, etc.) or process that induces cancer or increases its incidence.
Mutagenic or genotoxic agent (such as triglycidyl isocyanurate): a chemical product that increases the frequency of mutations in populations of cells and/or organisms.
Toxic for reproduction or reprotoxic agent: a chemical product (such as lead) that causes adverse effects on sexual function and fertility in adult men and women, as well as undesirable effects on the development of their offspring.
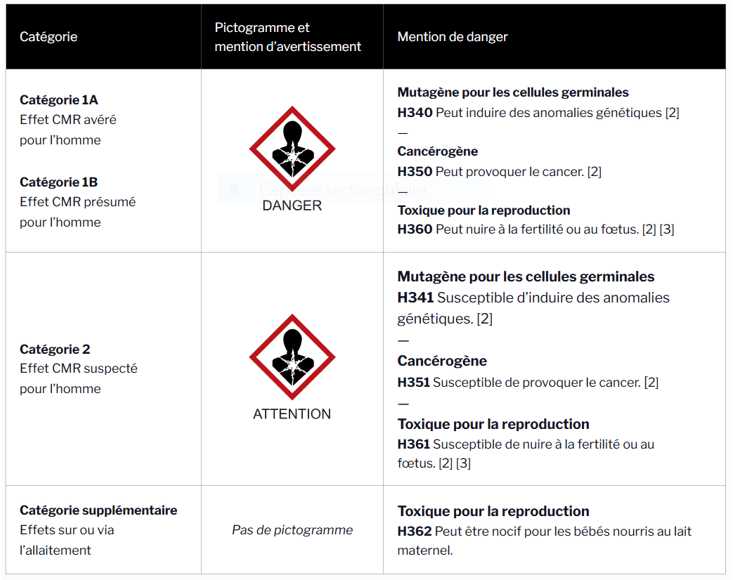
It should be noted that the classification and labeling of CMR substances and mixtures have evolved with the entry into force of the CLP regulation.
What is the Tolerable Daily Intake (TDI)? The TDI is the estimated amount of a chemical substance that can be ingested daily over a lifetime without presenting an appreciable risk to health. TDIs are expressed in terms of body weight, generally in milligrams or micrograms (of substance) per kilogram of body weight per day in the case of repeated exposure.
To assess safe use and exposure limits for these substances, Europe relies on several health safety agencies that can conduct or commission toxicological studies.
ECHA, the European Chemical Agency, was founded in 2007 in Helsinki. In collaboration with EU countries, ECHA analyzes chemical and toxicological data on substances to identify those potentially hazardous and to provide scientific opinions on risk management, for example by limiting their use or requiring prior authorization. ECHA’s scientific, independent, and high-quality opinions are submitted to the European Commission and EU Member States to guide actions and support final decisions.
EFSA, the European Food Safety Authority, was established in 2002 in accordance with European law, following a series of food crises. EFSA provides the scientific basis for laws and regulations aimed at protecting European consumers from food-related risks, from farm to fork. It delivers independent scientific advice and support to risk managers and EU policymakers in the fields of food and feed safety. EFSA also ensures independent and timely communication on risks and promotes scientific cooperation.
In the United States: the FDA, Food and Drug Administration.
The FDA is part of the U.S. Department of Health and Human Services. FDA-regulated products account for 20 cents of every dollar spent by American consumers. It is the oldest consumer protection agency of the U.S. federal government. For more than 100 years, it has been responsible for advancing public health by helping to accelerate innovations that make medical products more effective, safer, and more affordable, and by helping the public obtain accurate, science-based information to use medical products and food to maintain and improve their health.
2- HISTORY
In Europe
In 2004, Regulation (EC) No 1935/2004 was published. This is the framework regulation governing food contact materials, whose Article 3 states that:
“Materials and articles, including active and intelligent materials and articles, shall be manufactured in compliance with good manufacturing practices so that, under normal or foreseeable conditions of use, they do not transfer their constituents to food in quantities that could:
a) endanger human health, or
b) bring about an unacceptable change in the composition of the food, or
c) bring about a deterioration in the organoleptic characteristics thereof.”
This was followed by more specific regulations for each type of material, defining the rules, migration limits, and testing conditions required to validate compliance of packaging intended for food contact. To date, European harmonization has not yet been completed for all materials, such as metals. Local regulations must therefore also be taken into account.
In 2005, following an EFSA opinion, Regulation (EC) No 1895/2005 was published concerning certain epoxy derivatives in materials and articles intended to come into contact with food:
BADGE (3), derivatives of Bisphenol A in simple terms, are now subject to migration limits;
BFDGE (4), derivatives of Bisphenol F, and NOGE, novolac glycidyl ethers, have their use and/or presence in the manufacture of materials and articles prohibited.
In 2011, harmonization for plastic migrations was finalized with the publication of Regulation (EU) No 10/2011 on plastic materials and articles intended to come into contact with food.
Bisphenol A is limited to a specific migration of 0.6 mg/kg and is prohibited for use in baby bottles, cups, and containers intended for infants and young children.
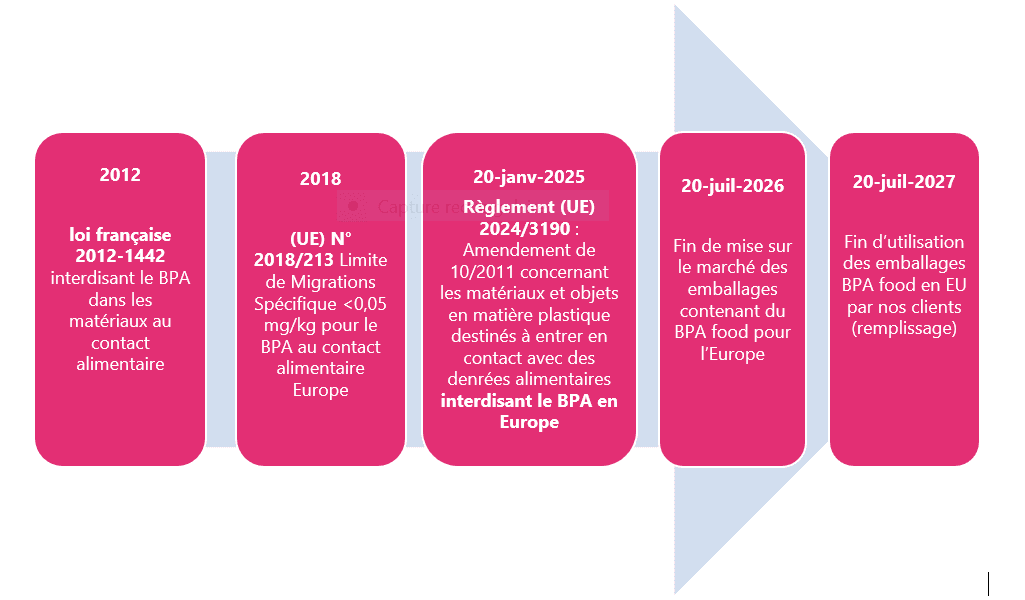
In 2012, France adopted Law No. 2012-1442, aimed at suspending the manufacture, import, export, and placing on the French market of any food packaging containing Bisphenol A. This publication was very important in Tournaire’s history, as it led us to develop products with BPA-free coatings, which we still produce today.
In 2018, Regulation (EU) 2018/213 revised the limitation of Bisphenol A (later repealed on 19/12/2024). Following a new EFSA opinion, the European Commission lowered the specific migration limit of Bisphenol A for food contact: it must now not exceed <0.05 mg/kg.
Latest EFSA opinion published on April 19, 2023, on BPA:
Dietary exposure to bisphenol A (BPA) poses a health concern for consumers of all ages, EFSA’s scientific experts concluded in a new re-evaluation.
Following a comprehensive assessment of scientific evidence and input from a public consultation, EFSA experts identified potentially harmful effects on the immune system.
The European Commission and national authorities then discussed appropriate regulatory measures to follow up on EFSA’s recommendations, leading to Regulation (EU) 2024/3190.
USA
National regulations concerning bisphenol A in children’s products and food contact materials:
Legislative bans on bisphenol A (BPA):
• 2012: The U.S. Food and Drug Administration (FDA) bans the use of BPA in baby bottles and spill-proof cups for children.
• 2013: The FDA bans the use of BPA in packaging materials for infant formula.
• 2015: Bisphenol A is listed under Proposition 65, List of Carcinogens or Reproductive Toxicants.
Asia-Pacific
Like many jurisdictions worldwide, several countries in this region restrict or ban the use of Bisphenol A in materials and articles intended for food contact. These include China, Malaysia (ban in baby bottles), South Korea (ban in all polycarbonate plastics intended for food contact), and Japan (migration limits in polycarbonate plastics). In Australia, some major retailers have voluntarily phased out the use of Bisphenol A in polycarbonate baby bottles.
3- LATEST EUROPEAN DEVELOPMENTS
In 2024, the latest amendment concerning Bisphenol A was published: Regulation (EU) 2024/3190, amending Regulation (EU) No 10/2011 and repealing Regulation (EU) 2018/213. It was adopted on December 19, 2024, and published in the Official Journal of the EU on December 31, 2024.
In 2025, on January 20, Regulation (EU) 2024/3190 entered into force, introducing the ban on BPA in Europe.
This regulation concerns the use of bisphenol A (BPA) and other bisphenols and bisphenol derivatives classified due to specific hazardous properties in certain materials and articles intended to come into contact with food. It applies to adhesives, rubbers, ion-exchange resins, plastics, printing inks, silicones, varnishes, and coatings.
Article 3 establishes the prohibition of BPA: the use of BPA and its salts in the manufacture of food contact materials referred to in Article 1(2), as well as the placing on the EU market of such materials and articles manufactured using BPA, is prohibited.
Compliance of packaging must be demonstrated by proving that BPA is not used in the manufacture of Food Contact Materials (FCMs) through supporting documentation for the Declaration of Compliance, and not necessarily through analytical testing.
Other hazardous bisphenols and bisphenol derivatives are also subject to restrictions.
In 2026, on July 20, the placing on the market of packaging containing BPA for food contact in Europe will end, in accordance with Regulation (EU) 2024/3190.
From July 20, 2026, it will be prohibited to place on the EU market metal packaging, lids, or closures with internal coatings or varnishes manufactured using BPA.
In 2027, on July 20, the use of packaging containing BPA for food contact (filling) will end, in accordance with Regulation (EU) 2024/3190.
In 2028, on January 20, the deadline for external coatings will take effect under Regulation (EU) 2024/3190.
From January 20, 2028, it will be prohibited to place on the EU market packaging or lids with external coatings or varnishes manufactured using BPA.
Packaging, lids, or closures manufactured using BPA and placed on the EU market before the above prohibition dates must be filled within 12 months following these dates and may then remain on the market until stocks of the filled packaging are exhausted.
4 - AT TOURNAIRE, THE CHOICE OF SAFETY: BPA-FREE PACKAGING
At Tournaire, we closely monitor these regulatory developments and choose safety of use, both for ourselves and for our customers. We ensure compliance through documentation provided by our suppliers, and we complement this with laboratory testing (internal or external) conducted by approved facilities. Our customers can therefore receive declarations of compliance and other certificates for our products.
Indeed, Article 411-1 of the French Consumer Code states:
“From the moment products are first placed on the market, they must comply with the applicable requirements relating to the safety and health of individuals, fair commercial practices, and consumer protection. The party responsible for placing a product on the market must therefore ensure that it complies with current regulations.” The entity placing the product on the market must therefore ensure the compliance of its packaging.
A key aspect of this compliance is the compatibility between the container and its contents—let’s look at the key points.
Ensuring the safety and health of individuals and consumer protection:
Whether for food, cosmetics, or other products, the product-packaging combination must be safe for human health under normal conditions of use.
The party placing the product on the market is responsible for the quality of the packaged product made available to the consumer throughout its shelf life. They are therefore responsible for container-content compatibility and for choosing the appropriate packaging system.
The packaging manufacturer, for its part, is responsible for ensuring that its packaging complies with the specifications defined by the client placing the product on the market.
Ensuring the quality of the packaged product throughout its lifetime:
The quality of a packaged product must be assessed from the design stage. This quality must be guaranteed throughout its life within the supply chain up to the end user. Product stability over time, under intended conditions of use, must be evaluated both in terms of organoleptic characteristics and potential interactions with the packaging.
Integrating container-content compatibility with the environment:
Packaging must comply with the essential requirements of European Directive 94/62/EC, soon to be replaced by the PPWR regulation (see our dedicated article). Compatibility must be assessed based on the strictly necessary use of packaging, while ensuring consumer acceptability and product protection.
5 - TOURNAIRE BPA-FREE SOLUTIONS
Matte natural finish (NM) or oxidized
Our aluminum containers without internal coating correspond to our “matte natural” finish. They can also undergo an additional oxidation treatment. In both cases, aluminum is the only material in contact with the product.
The aluminum raw materials and the very specific grades used to manufacture our containers (grades 1050A and 1070A) are among the purest forms of aluminum and comply with European standard EN 602 “Aluminium and aluminium alloys – wrought products – chemical composition of semi-finished products used for the manufacture of articles intended to come into contact with food.”
Our manufacturing process complies with the fundamental requirements of Regulation (EC) No 2023/2006 of December 22, 2006, on good manufacturing practices for materials and articles intended to come into contact with food, following a true quality organization (QA/QC), ISO 9001 certified, involving quality records, continuous traceability, and appropriate procedures.
Aluminum raw materials of grades 1050A and 1070A also comply with French regulations defined by the decree of August 27, 1987, relating to aluminum in contact with food.
The Food and Drug Administration (FDA) has historically issued favorable opinions for unalloyed aluminum metals in contact with food (assuming negligible migration into food).
Thus, our matte natural packaging, using near-pure aluminum and responsible manufacturing processes, is fully suitable for food contact. It is also used for other highly demanding applications, such as in pharmaceuticals, for packaging active drug substances.
However, our aluminum containers should not be used for acidic or salty foods in prolonged direct contact with aluminum. In such cases, TOURNAIRE can provide aluminum containers coated with an internal varnish.
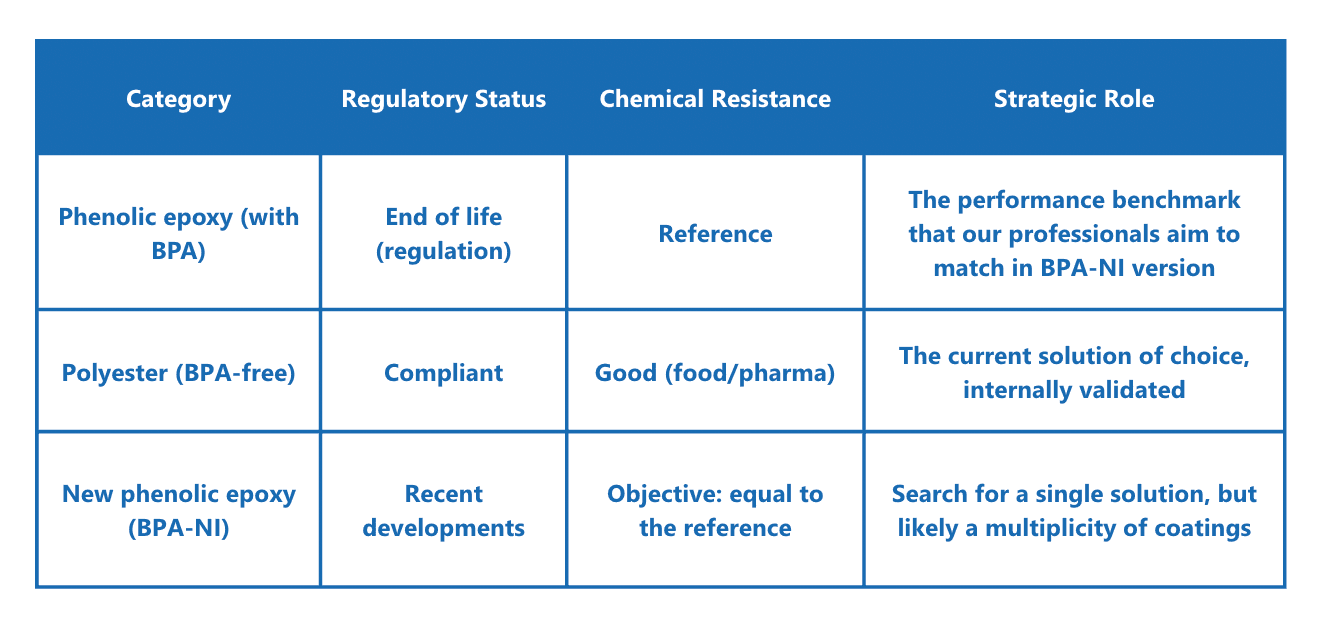
BPA-free varnish: VSB
The BPA-free varnish, called “VSB” internally (BPA-free polyester-based coating), has been in use in our workshops for over 10 years. It is a resin specifically designed for food packaging and is widely used in the beverage can industry, for example. Our technical expertise has enabled us to successfully deploy its use in our facilities, protecting the interior of our aluminum containers and expanding their applications for food contact.
This polyester-based coating contains no epoxy resin or epoxy novolac resin (NOGE) and can therefore be considered free of BADGE, BFDGE, and NOGE.
It is in our newest building, fully redesigned and dedicated to coating processes, that our VSB-finished containers are internally coated with this polyester varnish.
Testing and validation within the Tournaire laboratory
At Tournaire, we have chosen to meet this challenge without compromise by complying with BPA-free regulations while maintaining strong chemical resistance, ideally close to that of epoxy-phenolic coatings. To achieve this, we have, for years, validated internal protocols within our integrated testing laboratory.
Our modern equipment, particularly climate chambers, allows us to carry out all the necessary tests to validate our solutions, thus ensuring product performance and our customers’ success.
In summary, the solutions are:
● Depending on the type of container, a matte natural or oxidized version of our packaging;
● The VSB varnish, an existing and widely validated solution (with regulated migration for the food industry);
● New coating technologies, such as BPA-free epoxy coatings for certain highly aggressive applications.
The Laboratory as a Guarantee of Zero Compromise
The strength of former epoxy coatings, which unfortunately contained traces of BPA, was their robustness. The challenge is to reproduce this level of performance with BPA-free coating alternatives. This has already been our challenge with polyester coatings in recent years.
Choosing the right alternative is too complex to be left to chance. That is why our integrated testing laboratory is at the heart of our strategy: our expertise in coating chemistry enables us to precisely understand and quantify the performance of polyester coatings and new BPA-free resins when exposed to chemical aggression from a wide range of products, as well as to multiple regulatory constraints.
Our expertise is based on controlled internal coating processes and a dedicated testing laboratory to support our customers through these transitions—a true partnership. We guarantee the reliability of our results by being able to carry out all necessary tests, from A to Z. We particularly value collaboration with our clients and conduct double-blind testing campaigns to support better decision-making.
To demonstrate compliance, we rely on the control of our coating processes, internal testing based on our technical expertise, and in-depth knowledge of our clients’ products. Depending on the case, we may also rely on external laboratories for more complex testing, such as migration analysis. A tailored approach for a turnkey solution.
Accelerated Aging Protocol
Our protocol is rigorous and fully controlled by our technicians:
● In-line control and monitoring on our automated installations (T0): measurement of thickness, adhesion, curing, resistance, and initial porosity. Filling of packaging with reference products based on our experience or our clients’ products (thyme essential oil, geranium oil, automotive adhesive, various perfumes or flavorings, for example).
● Accelerated aging tests: aging at controlled temperatures (30°C to 40°C) and defined humidity, in monitored and recorded environments, with periodic checks of coating integrity to track progress (1, 3, 6, 9 months, and 1 year, etc.).
● Success criteria: zero defects and full physical integrity (perfect appearance, unchanged porosity), along with observation of the packaged product (validated by the client or external parties).
● Validation through regulatory migration testing for the food industry.
Regulatory Migration Testing: Approved External Laboratory
The main risks related to packaging choice and consumer safety are linked to interactions between the container and its contents. To measure these and verify the absence or limits of migration of the most critical substances, specific tests are required, as defined in Regulation (EU) No 10/2011.
There are also methods to predict substance migration using modeling software, based on data describing the packaging system, transfer properties of substances, material characteristics, and conditions of use. This approach is used as a decision-support tool during product development. The result is a curve showing the kinetics of substance migration into the contained product, expressed in mg of substance per kg of product, allowing prediction of substance levels over time.
At TOURNAIRE, we have chosen to perform global migration analyses to measure the transfer of non-volatile substances into simulants, in order to estimate possible changes in product composition. We also analyze specific migration of substances subject to usage restrictions (SML) in simulants or real products, using standardized or validated methods carried out by accredited laboratories, as required by Regulation (EU) No 10/2011.
These tests are performed by an external laboratory accredited under ISO 17025 and are available to our customers upon request. Based on these results, we also provide our clients with a Declaration of Compliance (DoC) established by ANIA (French National Association of Food Industries), specifically designed to facilitate the interpretation of test results and improve communication between clients and suppliers.
6 - CONCLUSION
The challenge of transitioning to BPA-free coatings and more broadly bisphenol-free solutions and supporting manufacturers through this change is complex.
Thanks to the control of our processes, our regulatory, development, and integrated laboratory teams, and above all our years of experience with a wide variety of substances across different applications, we are able to support our clients and define with them the best transition strategy based on their products and the applicable regulatory landscape.
In particular, we can determine the most appropriate testing strategy to ensure regulatory compliance today while guaranteeing future chemical excellence.
To make no compromises: let’s combine performance, high standards, and collaboration.
Sources and Resources
1: Product recalls in France, RappelConso statistics V1 – deprecated, https://data.economie.gouv.fr
2: https://fr.wikipedia.org/wiki/Bisphénol_A
3: bis(2,3-epoxypropyl) ether of 2,2-bis(4-hydroxyphenyl)propane, hereinafter referred to as “BADGE” (CAS No. 001675-54-3), and certain of its derivatives
4: bis(2,3-epoxypropyl) ethers of bis(hydroxyphenyl)methane, hereinafter referred to as “BFDGE” (CAS No. 039817-09-9)
Links to reference texts and websites
https://eur-lex.europa.eu/legal-content/FR/TXT/PDF/?uri=CELEX:32004R1935&qid=1757338787635
https://eur-lex.europa.eu/legal-content/FR/TXT/PDF/?uri=CELEX:32005R1895&qid=1757336792718
https://eur-lex.europa.eu/legal-content/FR/TXT/PDF/?uri=CELEX:32011R0010&qid=1757339206071
https://www.legifrance.gouv.fr/loda/id/JORFTEXT000026830015
https://eur-lex.europa.eu/legal-content/FR/TXT/?uri=CELEX%3A32018R0213&qid=1757339277182
https://eur-lex.europa.eu/legal-content/FR/TXT/PDF/?uri=OJ:L_202403190&qid=1757339314316
https://eur-lex.europa.eu/legal-content/FR/TXT/PDF/?uri=CELEX:32006R2023&qid=1758717421293
https://www.efsa.europa.eu/en/news/bisphenol-food-health-risk
https://www.efsa.europa.eu/en
https://echa.europa.eu/fr/home
https://www.fda.gov/
https://www.prc.cnrs.fr/reglementation/le-reglement-clp/
http://www.conseil-emballage.org/wp-content/uploads/2017/05/Compatibilité-contenant-contenu-Final.pdf
